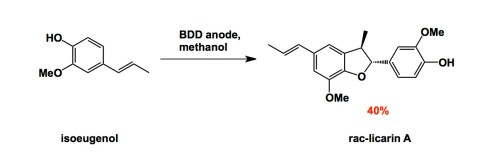
Electrosynthesis is one of my long-time passions, although my lab has not done it recently (but we shall…). I nonetheless pay attention to novel materials that people start to use these days. Here is one: boron-doped diamond. This material has been known to inorganic chemists, but recently the group of Nishiyama in Japan showed that anodic oxidation on the surface of boron-doped diamond leads to very effective formation of methoxy radicals. Learning from this fairly simple case, the authors expanded the method to the synthesis of a natural product, licarin.
http://onlinelibrary.wiley.com/doi/10.1002/anie.201200878/abstract
I am curious if this and other unusual electrode materials will find new applications in selective oxidation.
Keep in mind that there is, of course, reduction accompanying any electrochemical oxidation. Protons typically get reduced to hydrogen gas on platinum. People rarely draw this reaction, though (it is assumed). Another thing to remember is that ANODE is where oxidation takes place, whereas CATHODE is for reduction. That’s a simple rule to keep in mind.
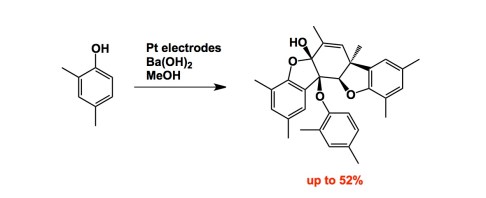
Since we are on the subject of oxidation, let’s increase complexity a bit… Here is Siegfrid Waldvogel’s excellent recent contribution to organic electrosynthesis. A good cumulative exam question, I must say! The increase in 3D complexity achieved in this synthesis is staggering:
http://onlinelibrary.wiley.com/doi/10.1002/anie.201006637/abstract