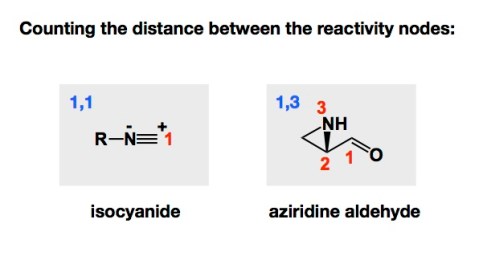
The Passerini and Ugi processes are two of the best-known multicomponent reactions that are based on the isocyanide functional group. In each of these processes, the isocyanide reacts with two carbonyl components. A carboxylic acid and an aldehyde are engaged in the Passerini process, while in the Ugi reaction it is the imine that reacts with the isocyanide instead of an aldehyde. When we started looking at amphoteric aziridine aldehydes with Ryan Hili back in 2006, we thought of aziridine aldehydes as 1,3-molecules based on the distance between the nucleophilic and electrophilic nodes of reactivity. This was a nice way to differentiate from isocyanides.
While aziridine aldehyde reactivity was in its infancy back in 2006, the isocyanide’s innate ability to react with the donor (eg aldehyde) and acceptor (eg acid) components had ample precedent in the Ugi and Passerini reactions I just talked about. Ryan and I felt that something must happen if you simply mix an aziridine aldehyde and an isocyanide. However, this simplest mode of reactivity has been elusive thus far. While we had plenty of luck with other processes, it has been kind of frustrating not to be able to find a precedent for what appears logical: mix isocyanide and aziridine aldehyde and get something cool. We are still trying to reduce this idea to practice. Unless my students correct me, aziridine aldehydes and isocyanides just stare at each other in solution. I still don’t understand why. Most likely Mother Nature is trying to teach me a lesson in kinetics here…
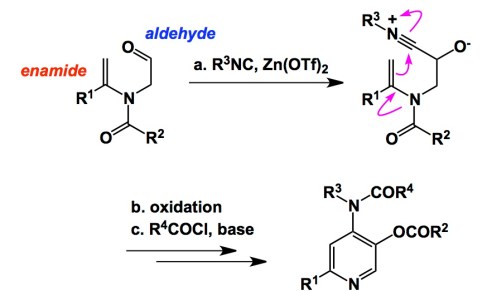
Along these lines, here is an interesting example that I really enjoyed reading about. The chemistry comes courtesy of Prof. Wang in Tsinghua University, China, and details the chemistry of enamide aldehydes that use their enamide portion in order to attack the isonitrilium ion, which is in turn created during the well-established isocyanide attack at the aldehyde (just take a page from the Passerini reaction!). The elements of amphoteric reactivity are on full display here. As a result of this well-orchestrated sequence, one gets access to polysubstituted pyridines. In the graphic below I am not attempting to illustrate all the gory details. While a couple of steps (oxidation and acylation) are left behind, the core of the process is present. Enamido aldehydes seem to be more than adequate to unleash the synthetic prowess of the isocyanide functionality…