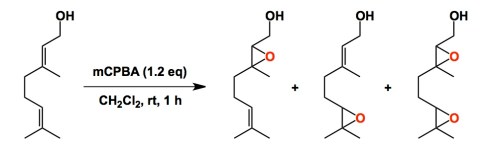
From a sheer pedagogical perspective, one of my all-time favorite papers is the well-crafted 2-page J. Chem. Ed. article by Tom Hoye and co-workers from the University of Minnesota. The goal of J. Chem. Ed. is to publish papers that serve as useful teaching tools. Hoye’s paper concerns with the epoxidation of geraniol. Sounds easy? The caveat is that the students have to perform this chemistry using 1mg of the starting material. The three epoxides derived from geraniol differ substantially in terms of their Rf. The students have to isolate and identify each product using conventional chromatography techniques. Each pure fraction is supposed to be characterized by 1H NMR. I know that the Chemistry Department at the University of Minnesota used this exercise as part of their teaching curriculum.
http://pubs.acs.org/doi/abs/10.1021/ed083p919
Organic synthesis is an experimental science and such small-scale experience is invaluable. According to Tom, less than 1% of new graduate students can do this with confidence. So, if you are a beginning (or an intermediate) synthetic chemist – my suggestion is that you give this reaction a shot in your spare time (using a 1 mg scale!). You do not need to tell the result to your advisor, but you will know if you need to put more work into developing better command of experimental techniques. According to Tom, the ability to perform in this test correlates really well with experimental acumen across the entire range of preparative techniques.

Pingback: Amphoteros – A Blogging Professor | Chemtips