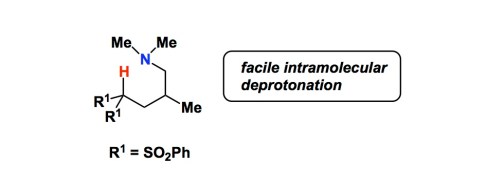
Synthetic organic chemists have always been awestruck by the remarkable rate enhancements on display in enzymatic reactions. One can list many different attributes possessed by nature’s catalysts. I want to talk about the most important one (in my view): control over solvation/desolvation. Is there anything synthetic that has this property? Menger has introduced the concept of spatiotemporal control, asserting that intramolecular reactions occur at enzyme-like rates when van der Waals contact distances between the reacting groups are too small to accommodate intervening solvent. For example, if two reactive groups are held in such a way that a water molecule can position itself between them, the groups are not considered to be in proximity. If we assume that water molecule is about 3Å in diameter, distances less than 3Å are expected to result in superfast intramolecular reactions. Here is an example. Enzymes are known to remove protons from fairly non-acidic CH groups using weakly basic histidine side chains (pKa of imidazole = 7). A synthetic model shown below has the CH-N distance of 2.34Å (which is less that 2.75Å sum of van der Waals radii of H and N). As a result, a fast intramolecular general-base-catalyzed proton exchange between the amino group and the methine proton takes place (measured through H/D exchange). This exchange is too fast to measure by NMR at -80 oC! In contrast, the intermolecular control is too slow to measure at 100 oC… The pKa’s of the amine conjugate acid and the CH bond marked below are 10 and 13.8, respectively. This example provides a “sneak peek” at what happens within hydrophobic pockets of enzyme active sites. Needless to say, synthetic chemists are keen on events that unfold on scales far greater than 3Å, which is why the so-called “enzyme models” never worked, do not work, and will never work. There’s just too much solvent around, once our overengineered molecules with all those cleverly crafted side chains are subjected to scrutiny. They might be decent catalysts, but they ain’t enzymes. My postdoctoral advisor, Barry Sharpless, once gave a talk entitled “The Secret Lives of Enzymes”. I think there is no secret, except for the unsurpassed control of solvation and desolvation.
amphoteros
forcing molecules to behave

Pingback: Amphoteros – A Blogging Professor | Chemtips