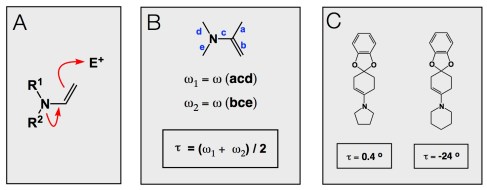
Proline has a number of distinct features. They range from its effect on the formation of turn structures in proteins to proline’s involvement in organocatalytic processes that are rooted in enamine chemistry (inset A). In our lab, we resort to proline when we run peptide macrocyclization: when used as an N-terminal residue in a linear peptide, proline plays an enabling role in the macrocyclization process. We have tried many other secondary amines, yet proline stands out. Interestingly, this observation mirrors organocatalysis in that proline’s core – its five-membered pyrrolidine ring – is a rather special enamine/iminium ion precursor. Notably, piperidine (a six-membered ring) pales in comparison as far as efficiency of iminium ion generation. The other day I was pondering over what makes proline so unique in our reactions and realized that I need to re-read some of the classic papers by Eschenmoser. His excellent Helvetica Chimica Acta work cited below describes X-ray structures of several enamines, including the two shown in inset C. The tau angle is what we want to focus on. Tau is a measure of nitrogen pyramidalization as it describes an average of two dihedrals marked by the small letters in inset B. According to this X-ray study, the pyrrolidine-derived enamine is more or less flat, whereas the piperidine congener is highly pyramidalized. This important crystallographic finding shows that pyrrolidine enamines have a shorter =C–N bond, leading to a higher C-N double bond character compared to their piperidine counterparts. The greater nucleophilicity of pyrrolidine enamines toward C-alkylation becomes clear and also explains why piperidine enamines are more prone to N-alkylation than pyrrolidine enamines.
http://onlinelibrary.wiley.com/doi/10.1002/hlca.19780610839/abstract