I am continuing my second year organic chemistry class, and although I am now covering NMR spectroscopy, the material of last week stirred some more “infrared memories”. I want to come back to the glory of this kind of spectroscopy and make it even more exciting… What if we want to see a situation where a unique reactivity insight is offered by way of analyzing an IR spectrum? This is different from the example I discussed in my last week’s post, in which the isocyanide functional group was distinguished from its cyanide counterpart through a shift in the wavenumber. This was more about functional group characterization. Now we want to shed light on reactivity preferences.
http://pubs.acs.org/doi/abs/10.1021/jo00078a014?journalCode=joceah
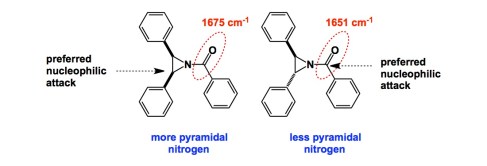
Above is a classic aziridine paper from Stamm and co-workers. Due to their substantial strain (about 27 kcal/mol), aziridines are different from other amines because aziridine nitrogen is much more pyramidal (which is to say that its barrier to inversion is high). Stamm’s experiments suggest that there is a marked difference between the chemistry of cis- and trans-acyl aziridines shown. You can see that the cis-isomer has a stronger C=O bond (reflected in the higher wavenumber). Interestingly, the cis-aziridine was found to preferentially react with nucleophiles (there were several nucleophiles and these details are beyond the scope of this post) at the carbonyl carbon, whereas the trans-aziridine preferentially reacted just like an epoxide. What might be the reason for this peculiar dichotomy? The IR spectra provide valuable clues. Apparently, the equalized steric environments of the top and bottom faces in the trans- case contribute to more efficient conjugation between the nitrogen lone pair and the carbonyl group. The trans-aziridine reacts as strained heterocycles normally do: the three-membered ring pops open. In contrast, in the cis-aziridine case, the acyl substituent prefers to occupy the less hindered face of the molecule. The nitrogen center is substantially more pyramidalized, which is reflected in less extensive conjugation and, hence, lower wavenumber. This explains why the carbonyl carbon is now preferentially attacked by the nucleophile. The Stamm study clearly shows the strong dependence of acyl aziridine reactivity on the nitrogen pyramid. In closing, IR can be really useful and full of insights.