I am sitting on my Oakville-bound train that is about to depart Union Station in Toronto. This winter has been super cold in our part of the country. It’s kind of funny because I have been hearing way less about global warming on the news. Weird, eh? I guess we’ll wait for a couple of months for people to start complaining. OK, I am being a troll.
Tonight I want to talk about the venerable Diels-Alder reaction. There is no need to praise it beyond the superstar status it already has. Instead of empty accolades, I will pay a facts-based tribute to this process and, in the spirit of recent discussions, try to poke at it.
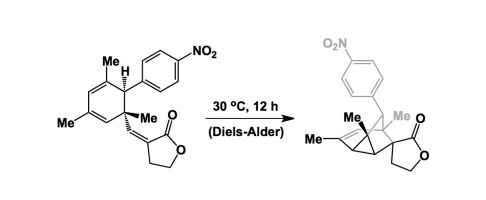
Are there 6-membered rings that cannot be made using this reaction? I can’t think of too many. The classroom value of Diels-Alder reaction is also undisputed: we beat this reaction to death when we teach frontier molecular orbitals (FMO) method, to the extent that some students leave our classes with an impression that this cycloaddition (and perhaps some electrocyclizations and sigmatropic reactions) defines the FMO theory in its entirety, which is not true at all. Nonetheless, this is still one of the most awe-inspiring reactions out there. To challenge its bulletproof status, one might want to subject Diels-Alder reaction to the limits of angular strain, hoping that that the cycloaddition might “crack”. Time and again, though, this resilient reaction has surprised us in most admirable ways. Take a look at one of my favorite papers on this subject. This is a study published by Dirk Trauner and Ken Houk some years ago. I would not have expected that imposing such severe strain on the 6-membered transition state would deliver any reasonable outcome. But the reaction works at 30 oC. There are some other interesting insights offered by this paper, so check it out. I think the lesson here might be that no matter how strain-crazy your idea might be, you should just give it a shot if it involves the magical 6p-electron transition state.
http://pubs.acs.org/doi/abs/10.1021/ja050135a
Now… Keeping up with the flavor of my recent post on heteroatom-heteroatom bonds, here is another viewpoint from my “vault of near-impossibles.” While there are countless examples of C-C and C-heteroatom bond formations using Diels-Alder reaction, it is interesting to note that heteroatom-heteroatom connections aren’t really made using this process. If you have a good example – please let me know. There is probably a fairly decent energetic argument against the transition state that produces a link between two heteroatoms (I should ask Ken Houk about this). Overall, I feel a bit better about showing that not everything is hunky-dory in the Diels-Alder bag of tricks. I will feel this way until you guys show me that I am wrong (or will you?).