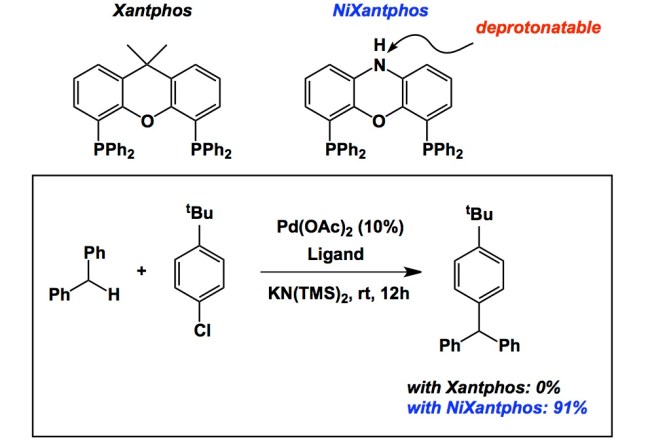
The correct choice of a ligand in transition metal catalysis is often the difference maker. Besides the steric and electronic parameters inherent to a given ligand structure, an interesting facet of structure/function relationship lies in the chemical modification of the ligand core during the reaction. This feature relates to a general question that is seldom addressed: how do you know that what you think is the ligand structure is actually responsible for the reaction under scrutiny? There are many examples where one can propose in situ modifications of the ligand core, yet one does not always monitor what happens to his/her catalyst during and after the reaction. I am sure there are many additional insights that might be obtained from such studies. There are, in fact, good examples showing the significance of in situ ligand modification. The one I want to cover today comes courtesy of Professor Pat Walsh at Penn. In his JACS report, Pat and his students describe the NiXantphos ligand that is deprotonated during the reaction. As a result, the authors observe facile oxidative addition of aryl chlorides to palladium coordinated to a bidantate ligand, which is unprecedented. Take a look below – there is no reaction with Xantphos, but 91% is obtained with NiXantphos. This is a striking example of how a “deprotonatable” ligand site plays the decisive role.
amphoteros
forcing molecules to behave