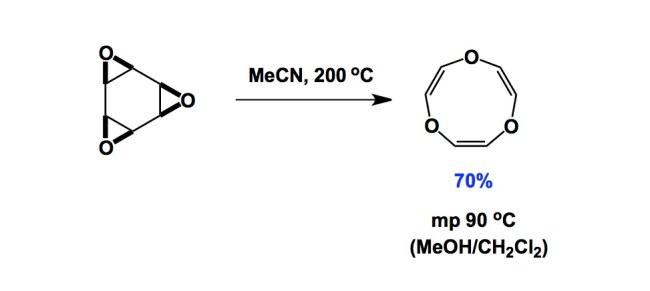
A lot of great science reported in the past was being pursued without an immediate application in mind. Earlier today, for reasons I cannot fully recall, I was thinking about a way of breaking the C-C bond of an epoxide. In doing so, I started reminiscing about some of Horst Prinzbach’s contributions to organic chemistry. Professor Prinzbach of Freiburg University has always been one of my scientific heroes, particularly after I met him some 20 years ago at one of the Loker Hydrocarbon Research Institute Symposia in Los Angeles. What stood out in my memory was Prinzbach’s paper published many years ago in Angewandte. In it, he and his students considered a thermally allowed [s2s + s2s + s2s] cycloreversion of a cyclic triepoxide that proceeded in good yields and selectivities. I am showing just one example below, but the scope is not limited only to epoxides. Mechanistically, this is one of those cases where it is easier to think of the microscopic reverse, rather than the forward process. You have to agree that the reaction shown below is not your mainstream epoxide chemistry. Sometimes I wish chemists could continue working on problems that do not have an immediate application. Such challenges are self-fulfilling and are about pushing the frontiers of fundamental structure and bonding, rather than seeking an immediate application. Unfortunately, the synthesis community crossed that Rubicon a long time ago and there appears to be no way back to the “science for the sake of science” type of research… I do encourage you to think about how you would go about making the triepoxide starting material shown below. As you might imagine, this is not that straightforward!
http://onlinelibrary.wiley.com/doi/10.1002/anie.197209421/abstract