As academic researchers strive to have their methods used in drug discovery, it is important to keep in mind that the reaction scope must include a large proportion of molecules that have a chance to have favourable drug-like properties. This is not always the case, and it is especially true when it comes to carbon-carbon bond forming reactions. Indeed, there are not many methods that accomplish carbon-carbon bond formation without turning synthetic precursors into “grease balls”. And what do you do when you are out of options? You turn to the other extreme and over-emphasize amide couplings, which creates molecules which are not exactly the stallions of drug discovery either.
The paper I intend to discuss is now a couple of years old (see the link below), but there are some practical items of substance in it that deserve attention. As rightly pointed out by Nakagawa and colleagues of Pfizer in their article, published methods tend to focus on excessively lipophilic model substrates, which is not what one wants to see in a molecule that is expected to be cell-permeable and have a chance to go through first-pass metabolism. In their efforts to find a reaction that accepts a broad range of building blocks, tolerates functional groups, and works well in the presence of moisture and air, the authors turned to BBC chemistry (Barluenga Boronic Coupling). This is a fascinating process that involves simple mixing of a boronic acid with a hydrazone. The reaction is reductive in nature and proceeds via an interesting mechanism. I was glad to see this process, originally published in 2009 in Nature Chemistry, in action. Despite the low isolated yields obtained by Nakagawa and colleagues, the simplicity of this protocol is attractive for rapidly assembling relevant molecules.
http://onlinelibrary.wiley.com/doi/10.1002/cmdc.201100339/abstract

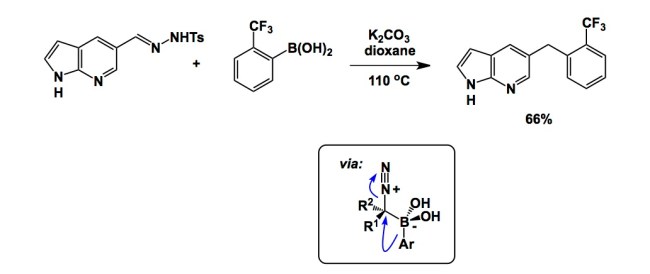
The reaction pathway, especially the Ar group migration from boron to the attached carbon to form the quatanary carbon centre reminds me a recent paper also in Nature Chem.
doi:10.1038/nchem.1971
Thanks!