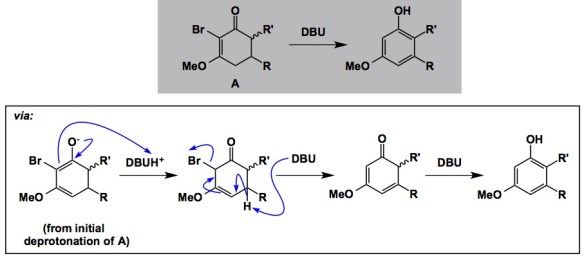
I have always been interested in mechanisms that lead to unusual elimination outcomes. Not that a decent leaving group is lacking in the reactions I am thinking about. On the contrary, a fairly middle-of-the road leaving group is, in fact, present. It’s just that the bond that is being broken is not the one you would typically expect to break. Consider the example that features an sp2 carbon-bound bromine (below). I learned about this interesting case from Professor Derrick Clive of the University of Alberta. I was in Edmonton over the past two days, attending a PhD exam of one of John Vederas’s students, Shaun McKinnie. The defense went really smoothly (it was an excellent thesis) and my long day culminated in a nice dinner with John and Shaun. But I kept thinking about the elimination sequence discovered in the Clive lab. You have to agree that it represents a peculiar reaction. The process features a tautomerization and aromaticity-driven removal of the sp2 carbon-bound bromine. While some might think that this is no miracle, especially once you consider how the double bond “dance” places bromine in a perfect position to eliminate in a vinylogous manner, I think this is the point here. The sequence offers a useful trick to make unusual phenols, among other things.