On a recent trip to Munich, I got acquainted with the research of Professor Hendrik Zipse. His mechanistic understanding of catalysis of alcohol silylation is both deep and educational. It serves as an instructive reminder of the reactivity/selectivity principle, which is something chemists relate to on an intuitive level. Not too long ago, Zipse and co-workers published a series of papers aimed at understanding the fundamental underpinnings of Corey’s classical silylation of alcohols. Zipse reminds us that DMF was the prescribed solvent in the original system. In this process, imidazole was used to mop up HCl, making TBS transfer one of the most familiar processes in organic chemistry. The question is whether or not the role of all components is crystal clear. It is now, but only after Zipse’s kinetic analysis. In brief, DMF is not your innocent by-stander. Its role is to form the active silylating argent, which is the Lewis acid/base pair shown below. Due to the high activity of this adduct, reactions in DMF (the original solvent from Corey’s 1972 paper) do not show impressive selectivity among primary, secondary, and tertiary alcohols. This is a very important finding. In contrast, if one stays away from DMF/imidazole mixture and runs silylations in dichloromethane along with DMAP and triethylamine, the selectivity is excellent. Improved reaction profile correlates with lower activity of the DMAP-derived active silyl transfer agent.
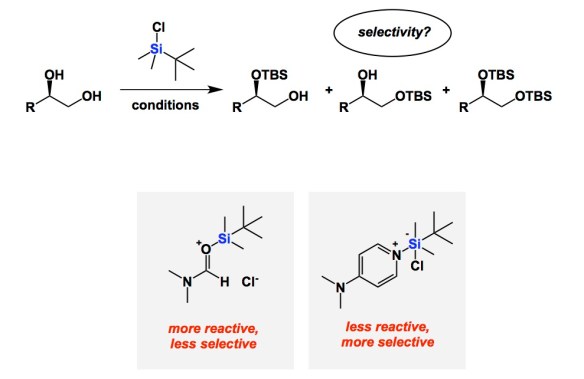

also to note is that imidazole used as a base in DMF/imidazole/TBSCl system is far from innocuous – you form N-TBS imidazole and imidazole hydrochloride in situ, and since imidazole has two nitrogens, most likely the protonated form of N-TBS imidazole is the actual silylating agent. (Also, the system works best if the react mix is highly concentrated, diluting it with DMF greatly slows the silylation – more than one would expect from a second order kinetics)
Imidazole/DMF system was developed to be nearly nonbasic, for sensitive substrates like alpha hydroxyketones. But for substrates that have no problem with base sensitivity and solubility, using TBS-Cl with DBU + DMAP in acetonitrile is the best choice for both primary and secondary alcohols, as well as lactam NH.
With monoprotected diols with one TBS group, their utility is limited to subsequent manipulations that do not involve a strong base – if you hit them NaH or MeLi, that silyl is really prone to migration to hydroxyl nearby, because of ease of pentacoordinate silicon formation. (I would rather go with something like less problematic like good old trityl group for such applications)
Good point. It is interesting how much stuff goes on among the seemingly trivial components of this system!
Hi Prof. Yudin, nice blog! I think you’re correct in that DMF does activate the Si, and off the top off my head, there’s a precedent for it – there’s some old papers from Prakash in the 2000’s where they were able to activate TMSCN and TMSCF3 with DMF.
Yes indeed. Thanks a lot!
One wonders then if these conditions could be used for activation of TMS-CF2H. Activating that molecule for -CF2H transfer is not quite as straighforward as with TMSCF3…
This is correct!