Boron-containing molecules are among the pillars of chemical reactivity. This is mainly due to the overwhelming embrace of the Suzuki cross-coupling, one of the easiest way to make C-C bonds. Over the years, it has been difficult to find other ways of coaxing boronic acids to participate in C-C bond formation. The paper by Tang and colleagues goes to show that simply raising temperature is a safe bet to discover new boron reactivity. What an irony, considering the sheer volume of research dedicated to getting organoboron compounds to react. Otherwise, how can anyone explain to me that alkenyl boronic acid nucleophiles have hitherto not been matched with alkyl halides? The reaction developed by Tang provides a new electrophilic partner for boronic acids, complementing the iminium ion reactivity captured by Petasis and colleagues 20 years ago. I find the process both interesting and useful. It takes place in toluene and requires about 80 oC to proceed. The success of this reaction is quite remarkable due to the absence of metal catalysts, which is why I tip my hat off to the authors.
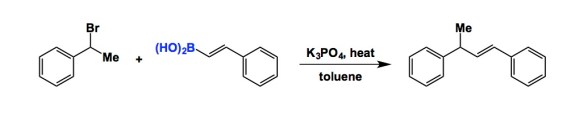

it would be interesting to find out if styrene-epoxides would be acceptable substrates for vinylboronic acid copling (Jacobsen asym epoxidation and chroacetophenone asym reduction with Noyori transfer hydrogenation are often complementary to asymetric dihydroxylation – they work for substrates where AD gives poor ees)
Also, it would be interesting to find out if O-alkylation of boronic acid is involved – i.e. if the product is formed by rearrangement of [Ar-CHMe-OB(beta-styryl)(OH)2 ](-)
Good point about epoxides, indeed. As far as the mechanistic details – I agree there are many unanswered questions, especially if one draws a parallel with the Petasis reaction and its reliance on O-B coordination.