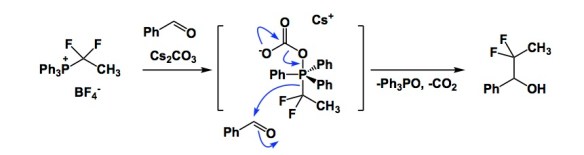
I have been intrigued by a series of papers from the lab of Professor Xiao of SIOC. The latest one just appeared in JOC and followed the Nat. Comm. report, which came out earlier this year. The reaction described in the JOC paper offers a route to difluoroethylated alcohols and amines. While it is easy to see why difluoroethylated molecules might be of interest in drug discovery programs, I am particularly intrigued by the mechanistic underpinnings of the Xiao process. Positively charged phosphonium cation is the source of the nucleophilic difluoroethyl group. The reaction appears to involve carbonate addition to phosphorous, at which points the “baton” is passed to the thermodynamics of the phosphorus-oxygen bond. This strong link is the reason why a nucleophile emerges from what is originally an electrophilic phosphorus component. By the way, this gives me an opportunity to lament, once again, on the ultimate origin of some common chemicals we take for granted. Take the venerable triphenylphosphine. Out of curiosity, I peeked into the Encyclopedia of Industrial Chemicals only to find out that this molecule is still produced from chlorobenene, sodium, and phosphorus tricholoride under intensive cooling. The corresponding oxide, which is made in almost all applications of triphenylphosphine in organic chemistry, is recycled by the likes of BASF using phosgene to first generate the Ph3PCl2 derivative, which is then reduced with aluminum. Wow. Talk about tracing common chemicals to their metallic origins (see my previous post).

less brutal (metal free) method of reducing PPh3O to PPh3 uses HSiCl3 but most companies do not even bother – I heard about large piles of PPh3O containing tons of waste oxide being just warehoused at Merck process plant.
By the way, in the difluoroethylation above, phosphonium behaves remarkably like TMS group actiwated with F(-) to form pentacoordinate Si center
It does indeed behave like TMS here, good point!
HSiCl3 is at least 5 times more expensive than PPh3. And what do you do with the product after the reduction? I wouldn’t bother to use it either.I think developing a metal catalyzed reduction is necessary.
This is true. This silane reduction is still a “lab-only” way. Not many people use this chemistry anyway other than in some complex (non-PPh3) ligand preps… Metal-catalyzed reduction will be a good way forward, I just hope that academics who work on this do not come up with some over-engineered solution that only technically qualifies as “catalysis”. They will surely need to mask the reduced P-center to prevent poisoning. And with what? With something like borane? So that it totally defeats the purpose…?
you probably can’t use boranes because BH3 forms ridiculously strong air-stable chromatography-compatible complex with phosphines. Breaking the complex takes cooking with several equivs of DABCO/
Also, Cl3SiH as a commodity chemical is surprisingly cheap in bulk (it is used for making solar panels) though when you buy lab quantity from Aldrich you pay all kinds of handling, packaging and hazard fees due to the fact that it is low-boiling highly toxic flammable liquid.
I agree. This is why I don’t think D.Stephan’s way won’t be as good as it seems. It is difficult to think of a catalysis with an early metal since you will hit another thermodynamic sink (M=O). Maybe some late metals like Ni, Cu will work if you don’t use great amounts of expensive reagents. Or less oxophilic main group elements will work (indium??). In the end, it all comes down to the cost I guess. You really have to use cheap reagents, or small amounts to make use of the system. Very difficult task but high reward in the end.
Thanks. I was actually not referring to Doug’s work. He has this amazing use of phosphine-borane adducts in catalysis of many reactions. But this is different. I was referring to the fact that phosphine-borane adducts (known for decades) have always been considered in efforts to tame phosporous. These BH3 adducts have been on the market for a very long time and have been used in stoichiometric quantities…
It is possible that there is a solution out there among the metals you mentioned though!