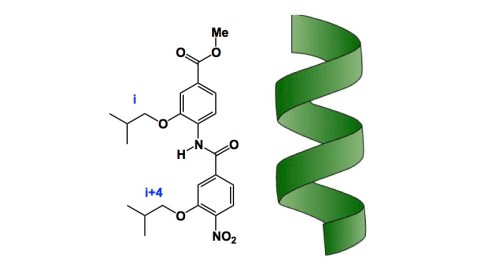
Who says that molecules have to be complicated in order to effectively interfere with protein-protein interactions in cells? Here is a fascinating recent example of a small molecule developed in the lab of Professor Ahn at the UT Southwestern, Dallas, Texas. This paper, published in Nature Communications by Ahn and his collaborator Ganesh Raj, is a lesson to all of us who think that mimicry of complex epitopes using small molecules is a lofty goal. Well, it certainly is a lofty one, but it is also reachable! This and other examples (Hamilton’s great work on polyaromatic molecules comes to mind) exemplify the state of the art in reductionist systems feasible with smart design. Look at the helical pitch (below) and its mimicry by a trivial amide compound. I am willing to bet there is a lot of completely unexplored molecules that are also small, perhaps a bit more architecturally advanced than the one shown, yet capable of interrogating non-helical epitopes, including disordered ones. In Ahn’s example, the IC50 of 40nm was achieved in efforts to disrupt specific protein-protein interactions involving LXXLL motifs. The use of such simple molecules in targeting androgen receptor-coreceptor interactions has been demonstrated to have clinical value. Kudos to Ahn and co-workers who designed these wonderful molecules!
The most important lesson I learned from my mentor, Professor Barry Sharpless of Scripps, is that complex problems do not require complex solutions (as opposed to what many people in synthesis preach). The present case certainly underscores this notion.
http://www.nature.com/ncomms/journal/v4/n5/full/ncomms2912.html