Here’s my Monday, October 7, 2013’s two cool facts about boron heterocycles.
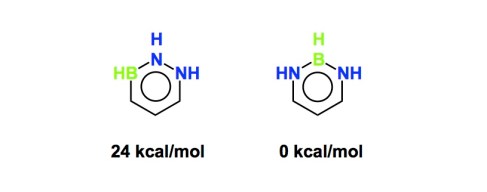
First of all, boron is tricky and it is not always obvious which factors control stability. Take, for instance, a recent theoretical JACS paper by Professor Roald Hoffmann of Cornell University. The manuscript contains a lot of very interesting data on boron heterocycles and is, in many ways, a call to arms for those who are interested in this subject. However, how would you predict the whopping 24 kcal/mol difference in stability (calculated relative energies) between the following two molecules? I realize they are not the same, but 24 kcal/mol? This means that NBN motifs are special…
A lot of really imaginative work in the area of aromatic boron heterocycles has been done by Prof. Liu of Boston College (formerly of the University of Oregon). I am not going to say anything about his nice synthesis of aromatic boron-containing heterocycles. Instead, I will focus on properties and showcase a co-crystal structure between azaborine and T4 lysozyme. The hydrophobic cavity of this enzyme accepted azaborine, a view of which I made using PyMol (pdb code 3hh3, below). It can be clearly seen that the azaborine molecule binds in two conformations which relate to each other by way of a “flip”. Given the amount of aryl groups in biologically active molecules, these boron heterocycles are certainly interesting! How about using them as fragments? Someone should…
http://onlinelibrary.wiley.com/doi/10.1002/ange.200903390/abstract