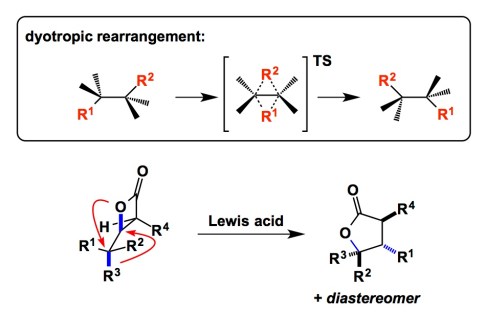
Nowadays, when one talks about discovering new reactions, one quickly realizes that there’s not a whole lot of fundamentally new elementary processes that remain unknown. As a result, novel reactions tend to be “composed” of different permutations of well-known elementary processes. Every now and then a mechanistically distinct transformation pops up. I keep an eye on reactions of that sort. Daniel Romo’s elegant experimental work coupled with Dean Tantillo’s theoretical approach provide a glimpse at some useful, yet fairly uncommon, types of reactions – the so-called dyotropic processes (see the graphic below). In sigmatropic rearrangements, a pi/sigma-system undergoes a transformation that results in a net translocation of one sigma bond and concomitant shift of the pi-system. In contrast, dyotropic rearrangements describe simultaneous migration of two sigma-bonds. Evidence in the JACS report cited below suggests that, depending on the nature of the Lewis acid, a concerted or stepwise mechanism takes hold. While reactions of this kind have been known for some time, they have not reached the mainstream of synthesis. I think there is a lot of room for reaction discovery using this mode of reactivity in the context of complex electrophiles.
http://pubs.acs.org/stoken/beta/select/abs/10.1021/ja303414a