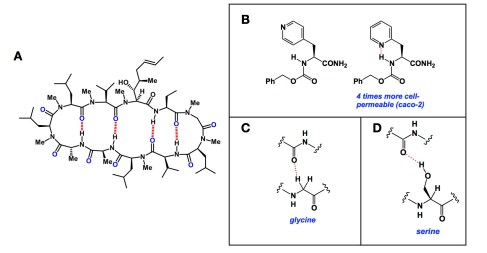
I was under the weather last night, so there was no chance to publish my Friday post. But I am back now, sitting in our living room and marveling at the copious amount of snow that has fell upon us over the past 24 hours. I think it is easily 25cm, although I have not heard the official forecast. Now… Hydrogen bonds hold together the snowflakes I am looking at. The impressive cooperativity of these weak forces is responsible for the snowflake formation. In a totally unrelated domain, the cellular permeability of peptides and amino acid-derived small molecules is also related to hydrogen bonds. Peptides are typically awful in their ability to traverse non-polar cellular membranes, save for a few exceptions. Cyclosporine A (shown in inset A below) is among the better ones. The network of hydrogen bonds that you see provides for a fairly lipophilic conformation of this molecule and helps it go through cellular membranes by passive diffusion. Well, at least this is what people think now (you never know when we will all flip our minds upon discovery of a predominantly active pathway that involves a protein transporter for cyclosporine A). The lessons offered by the internally satisfied hydrogen bonds here extend to other areas. The inset B below shows a marvelous case from a paper by Jacobson and colleagues: http://pubs.acs.org/doi/abs/10.1021/jm201634q. The two molecules clearly have identical polar surface areas, yet the one on the right is 4 times more cell-permeable by virtue of the hydrogen bond marked in red.
So what should those, who want to design cyclic peptides with improved cellular permeability, take from all of this? I think the main lesson is that we need to continue our hard-target search for hydrogen bond pairs and they do NOT need to be restricted to what you see in the case of cyclosporine. The classical NH-O hydrogen bonds are cool, but we need to go way beyond that. Here is a paper that talks about glycine’s ability to engage its alpha C-H bonds in polar contacts (inset C) that lead to stabilization of inter-helical motifs in proteins: http://www.pnas.org/content/98/16/9056. Does this mean that we want to see more glycines in cyclic peptides in order to make them have better chances of being cell-permeable? I am not sure. But I do know that glycine has two alpha C-H bonds and this is the reason why the likelihood of forming the “right” connection is simply higher for glycine. It follows that perhaps d-amino acids should be considered more often if these unusual hydrogen bond motifs are to be captured. Lastly, I will present another case that is well familiar to protein chemists – that of serine (inset D). Here is a paper showing that serine is a residue par excellence in “reaching over” and forming hydrogen bonds using its OH group: http://www.biomedcentral.com/1471-2148/10/161. Someone has to take all of this protein chemistry knowledge and translate it into cyclic peptides. We may then indeed start to see trends that will emerge from “unusual suspects” for hydrogen bond formation in macrocycles. I do want to end by saying that these less common hydrogen bonds are fairly weak, but they sure have their place under the sun!