A week or so ago I had a discussion with one of my former students about what constitutes the most important skill to acquire in preparative synthetic chemistry. This is a complex and multilayered question simply because we have so many methods in our disposal. The list grows as new ways of interrogating molecules become accessible. Sometimes I think that we have way more than we need, to the point that instrumental cornucopia stifles the development of solid synthetic skills. When I was in graduate school there was a visiting scientist in our lab who was having a difficult time coping with his daily lab duties after our GC/MS machines broke down. This is an unfortunate situation that speaks to the “instrument addiction”. What did synthetic chemists do 40 years ago? They had less instrumentation, but I bet they were better prepared to do experimental science because they had no choice but to be resourceful. I do have some extreme views in this regard, as my students would testify. On a number of occasions I mentioned that we should just get rid of our two LC/MS systems because they provide too many false positives as well as false negatives (the worst possible combination!).
Now let’s get back to what I would consider to be the most important skill to acquire. I suppose that once an experimentalist knows how to efficiently isolate his/her products and set up reactions, everything comes down to following the reaction progress and developing top-notch observation skills. To me, it is all about TLC (thin-layer chromatography), which is the most accessible and rapid analytical tool possible. I think that we need to develop behaviours that are close to obsessive-compulsive in regards to how often we run TLC of our reactions and how early we apply our first TLC spot in a given case.
http://pubs.acs.org/doi/abs/10.1021/ja0576379
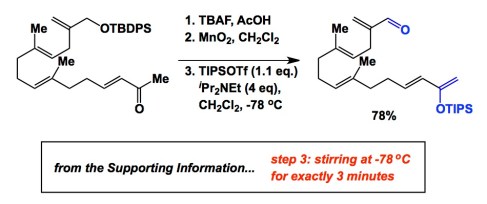
How often should one take a TLC measurement and when is a good time to take the first one? This is case-dependent and I am going to provide an example that strikes at the heart of the problem and makes us think (especially those of us who routinely deal with fairly unstable molecules). The reaction above comes from a total synthesis published by E. J. Corey and Scott Snyder several years ago. Scott told me about this example at a conference. If you run your first TLC of step 3 at 4 minutes after its start, it is too late and you will not get much product because the molecule is too unstable under the reaction conditions. However, once the reaction has been properly worked up after 3 minutes, the target molecule is isolable. I am sure there are many more examples like this out there and they all serve an important lesson. If you are a student, imagine that you are trying to make a sensitive intermediate and you are screening conditions that result in the infamous “messy TLC” description in your lab book. When did you take your first TLC?