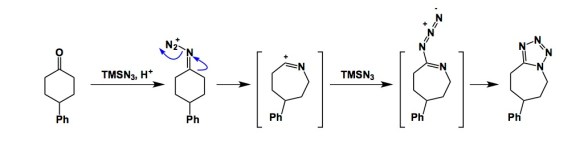
Not long ago, I talked about the power of the Aubé-Schmidt reaction in the synthesis of unusual amide structures. I just saw a JOC report by the Aubé lab that details a rather unusual outcome of this process when TMSN3 is made to react with ketones in the presence of triflic acid (TfOH) promoter. In the course of this reaction, tetrazole formation turns out to be the predominant pathway. This outcomes stands in contrast to established protocols in which one typically expects to see lactams or amides through formal NH insertion into the C-C(O) bond. This mechanistically distinct process hinges on the prior discovery that 1,1,1,3,3,3-hexafluoroisopropanol (HFIP) acts as the catalyst and reduces the number of equivalents of acid needed for good conversion in the Aubé-Schmidt reaction. In the most recent embodiment, the sequence of iminium ion formation / isonitrilium ion formation / ring-expansion, is observed. To sum up, the reaction between azides and carbonyl compounds continues to have an enduring impact on the synthesis of small molecules.