Here is an interesting and teachable case that seamlessly merges the elements of biosynthesis and fundamentally important classic organic chemistry. Liu and colleagues report indolenine 1 as the “cryptically conserved” intermediate in the biosynthesis of hapalindole-type alkaloids. It all starts with the magnesium-dependent formation of 3-geranyl 3-isocyanovinyl indolenine from cis-indolyl vinyl isonitrile and geranyl pyrophosphate (this process is enzymatically catalyzed by AmbP1). What ensues is an interesting Cope/aza-Prins cascade, which ultimately leads to divergent pathways to hapalindole-type alkaloids. Besides an intriguing biosynthetic route, this work is important as it opens doors for understanding the molecular basis of the metal dependency of prenyltransferases.
Hapalindole alkaloids have received their fair share of attention in the synthetic community, but only Ang Li et al’s work (http://onlinelibrary.wiley.com/doi/10.1002/anie.201406626/abstract) foreshadowed the Cope/aza-Mannich biosynthesis put forth by Liu. While it is nice to read such stories, I am not suggesting that the present case is an exception. The most famous case of “foreshadowing” is perhaps Stork’s classic work on enamines, which had appeared way before type I aldolase mechanism was elucidated.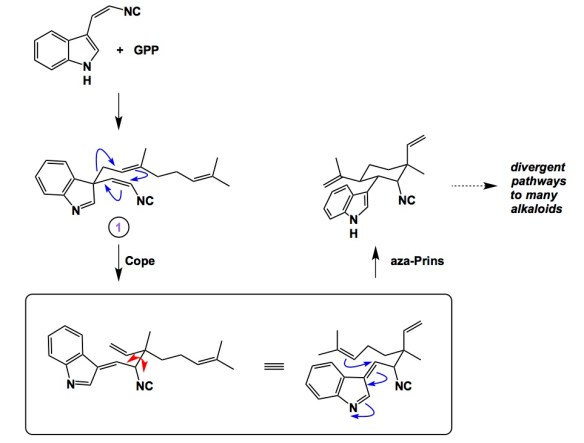
http://pubs.rsc.org/en/content/articlehtml/2016/cc/c5cc10060g
