Deciphering the structures of random reaction by-products is rarely on top of our “to do” list. This avoidance is understandable because constant pressure to move toward the stated goal makes us focused, which is both good and bad. Noticing reaction by-products is how one makes important breakthroughs, yet the chances of finding something truly spectacular in the “noise” are not in one’s favor.
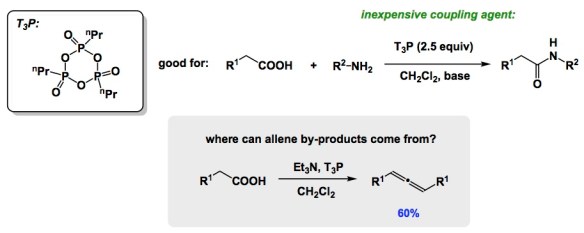
As a research advisor, I love learning about crude NMRs. But I am also aware that our resources and bandwidth to pay attention to what comes our way are not limitless. This inevitably leads to (un)healthy skepticism. A recent paper in Organic Process Research and Development stopped in my tracks the other day because I clearly recall hearing about allene formation (by NMR) in one of our peptide coupling reactions. This observation was made while analyzing the reaction by-products and I dismissed it as “OK, the NMR might suggest there is something like an allene but it is unlikely, so let’s move on”. In the case of Knapp and colleagues, the acid used in the coupling process did lead to an allene. There is even a crystal structure of the product reported (it is a complex structure, so I am avoiding it here). The mechanism of allene formation is cool and involves ketene formation. I am not saying that definitive identification of a similar structure in our reaction several years ago would have affected the line of research we had been pursuing at that time, but I just wish we all had more time to dwell on what’s brewing in our flasks. Incidentally, I think it was BASF that has been pushing T3P as a fairly inexpensive reagent for amino acid coupling. It is a nice little molecule.

Indeed you never know where the next new thing will come out of. Incidentally, we’ve had a couple of papers out by analysing unexpected products and by-products 🙂
DOI: 10.1039/C6CP04321F
DOI: 10.1016/j.tet.2016.05.058
Nice work! I like what happened in the sulphur system…
I am bookmarking this post so that I can come back and link my article when we publish. I did structurally characterize an interesting new X-Y bond formation example from one of my totally unrelated reactions.