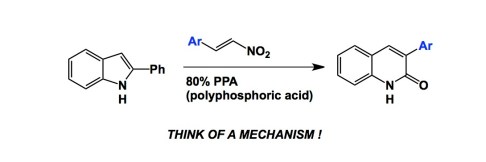
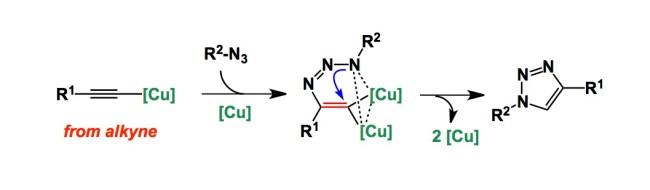
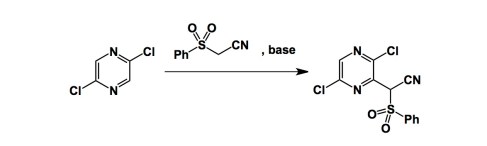
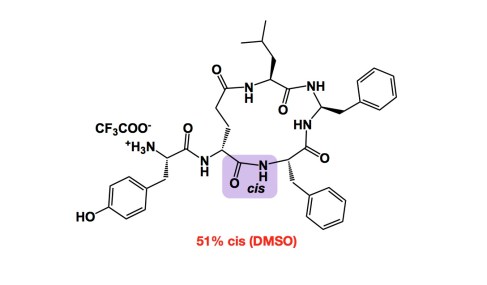
Had anyone told me 5 years ago that my lab would be heavily engaged in peptide chemistry, I would not believe a word… Fast forward to 2013 and here I am, together with my man Ved Srivastava of GlaxoSmithKline, organizing the next American Peptide Symposium. What a turnaround.
Here is an early-bird invitation to everyone who is interested in peptide science. Come to Orlando in June, 2015! We will have a splendid conference prepared for you. Our logo is below and a link to the website is forthcoming. So, bring your significant others and have some fun in Orlando with us. Besides the scientific program, Ved and I will work hard to ensure that entertainment is taken care of. I will bet that there will be some nice Cuban food, cigars, and so on. This past APS meeting was held on the Big Island of Hawaii and it was awesome (kudos to Marcey Waters and David Lawrence). The only downside was that it was a bit too far and many people could not make it (especially industry folks who just could not convince their upper management that there is science in Hawaii). Orlando, on the other hand, is strategically placed to embrace both the European and American contingents.
I will also start spilling the beans about some special features we have in store for your amusement… As part of the conference, we are preparing a cool new 2-hour section called “rapid-fire”. At the beginning of the conference, the attendees will be given a chance to email the organizing committee two slides with their latest results. Of course, you need to consider the pros and cons of this disclosure. We will then select speakers and give them 5 minutes each in the rapid-fire section. 2 slides, that’s it! This is going to be a new mechanism to give floor to young researchers such as students and postdocs who may have a cool result, but perhaps not enough material for a full-scale oral presentation. This lack of a full story is often the reason for one’s reluctance to submit an abstract for a talk. The rapid-fire section will not have a pre-announced program. We plan to call the chosen speakers from the audience during the allotted time. These are our plans and we’ll see how they materialize. We certainly hope they do!