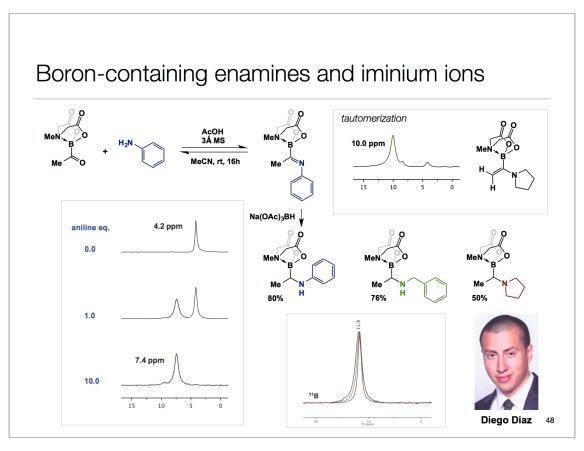
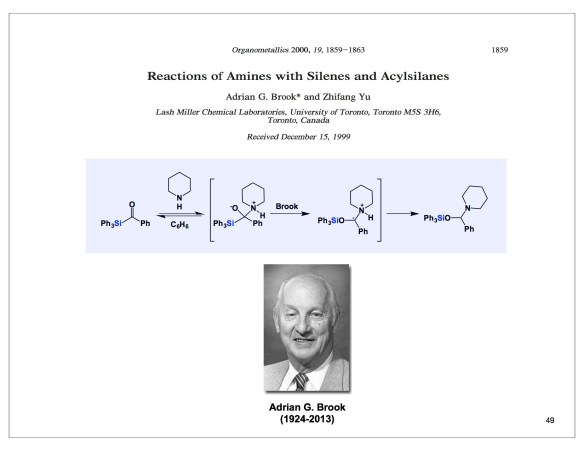
Now that the fall semester is finally here, I will hopefully have more time to write my posts. Today is all about Diego Diaz and the cool chemistry we recently published in Angewandte (http://onlinelibrary.wiley.com/doi/10.1002/anie.201605754/abstract). Diego did his undergraduate work with Patrick Gunning and came to do PhD in my lab in the Fall of 2014. He quickly developed a keen interest in placing boron within peptides, gave it all he had, and came up with what I think is the best way to incorporate boron into amino acids and related structures. You might wonder why and I could name a few applications: from cross-coupling all the way to hydrolase inhibition. But I refuse to talk about any of this tonight because the ultimate target of our research endeavors is to understand the basic reactivity of organic molecules. In this regard, Diego’s sigma-loaded iminium ions stand out. We have not only characterized them, but we have also employed them in several reactions, including one of my favorite ways of linking molecules – by way of reductive amination. Below are some of the details. Using two slides from a lecture I gave in Halle (Germany) 10 days ago, I show Diego’s NMR data. With respect and admiration, I also pay a tribute to my late colleague, Professor Adrian Brook. The Organometallics paper you see was Adrian’s last contribution to chemistry. It is fitting that this manuscript details an attempt to make imines from Adrian’s acyl silanes. As we all know, this is not possible with silicon because of the Si-heteroatom bond strength, which triggers migration (Brook rearrangement). In our case, we do not have evidence of migratory processes, which is due to the carefully chosen tetrahedral environment around boron. This is amusing, given the fact that boron, not unlike silicon, loves oxygen.
As “luck” would have it, right about time when Diego’s chemistry entered its high gear, he is moving to Vertex in Montreal, but thankfully only for three months (this is one of those industrial experience shindigs). Let’s see what he will be able to accomplish by Christmas. I hope to be able to disseminate the non-confidential part of it. For now, I am really happy about the facility with which we can “smuggle” boron into the structures of bioactive molecules. Thanks Diego.