A couple of weeks ago I gave a talk at the Scripps Research Institute. My visit was superbly organized by Professor Phil Dawson, for which I am thankful. The highlight for me was to interact with my mentor, Professor Barry Sharpless, who celebrated his 75th birthday earlier this year. We have not seen each other for a while, but we picked back up as if we never parted. As much as ever, I felt energized after our long meeting, where we shared some memories from days past. Barry told me about his new passion, which is the SuFEx reaction for stitching molecules together. SuFEx is a “click” process, with Barry’s well-known stamp written all over it. There are some spectacular new papers about to come out of the Sharpless lab on the subject, but you can take a look at this published one for starters: http://onlinelibrary.wiley.com/doi/10.1002/anie.201309399/abstract. Speaking of Barry’s other famously known click transformation, I received a sweet present – a set of copper balls, in association with the copper catalysts used in the azide-alkyne cycloaddition. Barry told me: “Andrei, now you need to shine your new balls”. By the way, these things are heavy, about a pound or so each. The most memorable part of this trip, though, came during Q & A session after my talk. A good chunk of the lecture was related to one of my lab’s current interests, namely the relationship between hydrolase inhibitor design and boron / oxygen reversible interactions (we have an interesting paper with Professor Ben Cravatt’s lab that will be sent out soon). Toward the end, Barry made some insightful remarks related to the boron work and one of them has stuck with me. On the pervasive coordination of oxygen to boron in some of our systems, Barry quipped: “…these two deserve each other”. What can I say? This was vintage Barry.
Monthly Archives: December 2016
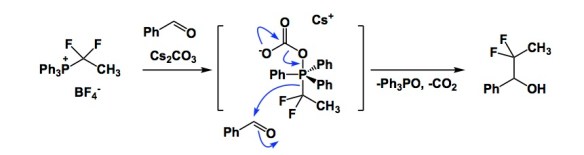
A nice way to transfer difluoroethyl groups
I have been intrigued by a series of papers from the lab of Professor Xiao of SIOC. The latest one just appeared in JOC and followed the Nat. Comm. report, which came out earlier this year. The reaction described in the JOC paper offers a route to difluoroethylated alcohols and amines. While it is easy to see why difluoroethylated molecules might be of interest in drug discovery programs, I am particularly intrigued by the mechanistic underpinnings of the Xiao process. Positively charged phosphonium cation is the source of the nucleophilic difluoroethyl group. The reaction appears to involve carbonate addition to phosphorous, at which points the “baton” is passed to the thermodynamics of the phosphorus-oxygen bond. This strong link is the reason why a nucleophile emerges from what is originally an electrophilic phosphorus component. By the way, this gives me an opportunity to lament, once again, on the ultimate origin of some common chemicals we take for granted. Take the venerable triphenylphosphine. Out of curiosity, I peeked into the Encyclopedia of Industrial Chemicals only to find out that this molecule is still produced from chlorobenene, sodium, and phosphorus tricholoride under intensive cooling. The corresponding oxide, which is made in almost all applications of triphenylphosphine in organic chemistry, is recycled by the likes of BASF using phosgene to first generate the Ph3PCl2 derivative, which is then reduced with aluminum. Wow. Talk about tracing common chemicals to their metallic origins (see my previous post).
Rosie Ruiz chemistry
As 2016 is slowly winding up, let’s turn to Scopus and see what’s been cooking in this chemistry universe of ours. Tonight we will take the unfortunate misnomer “metal-free”. It turns out that in 2016 the synthetic community has churned out a whooping 1391 papers containing this topic. The vast majority of these papers are interesting and potentially useful. But this is despite being labeled “metal-free”, not due to some features of this dubious concept. This whole thing reminds me of Rosie Ruiz, who won the 84th Boston Marathon in 1980 in the female category. Her title was later taken away when it was uncovered that Rosie took the subway for a good chunk of that run. I liken many of the metal-free approaches to Rosie’s feat. Thankfully, these papers expose a huge gap in chemistry education: we do not provide our students with the origins of industrial chemicals. How many of us know how common components (such as benzaldehyde, pyridine, aniline, etc) of “metal-free” reactions are made in industry? Armed with this information, we might be able to better appreciate that the heavy lifting is often done early, using metal-based chemistry that is far less glorious than the picture painted later by those “metal-free” routes. In this age of sustainability, I always want to keep in mind that synthesis is not a sprint but a gruelling marathon.
