If I were to run a poll, asking the practitioners of cyclizations about their most commonly dreaded side reactions, I am sure the answer will be clear: oligomerization and polymerization will be close to the top of that list. Entropically, this is to be expected of a bifunctional intermediate, especially when it comes to macrocyclization reactions where entropy is really not our ally. I was interested in seeing a reaction shown below. Not long ago, this process was reported by Kitamura and co-workers in Organic Letters. I find this palladium-catalysed cyclization interesting because it represents a somewhat odd outcome: this is not what you might consider an oligomerization, yet this is also far from a simple annulation. I chose one of the better examples. There are indeed mixtures of products in other entries reported in this work. But still, this “in-between” business when the system is not reaching the oligomeric abyss is interesting and is undoubtedly governed by some good rationale – so you should look at the mechanism.
Monthly Archives: June 2014
Things that should not work
When we learn the fundamentals of organic chemistry, we tend to receive trivialized material where many details are omitted for clarity. I think this is not only essential, but is also critical to the establishment of a solid chemistry foundation. The trouble is that we leave our students in the dark when it comes to gaps. I suppose graduate school is expected to take care of those gaps… Let’s look at Sn2 chemistry at sp2 centers. This should not work, yet Professor Narasaka has spent a good portion of his career perfecting these kinds of reactions. Below is a reference to a review he has written and an example of a well-known Sn2 substitution at a hydroxylamine nitrogen center. The fact that the syn-form does not lead to any product formation supports the feasibility of backside attack (and so do many other detailed mechanistic studies). Another example I include comes from Merck and shows an awesome and scalable way to make pyrrolidines. This high-yielding process employs an alkyl halide, which is a “no-no” when we teach undergraduate chemistry. This is due to overalkylation nightmares (“since this cannot work, let me tell you about the Gabriel synthesis…”). By the way, if you take a look at the reference, you will note that this chemistry works on a multi-kilogram scale and delivers fantastic yields. You can also use similar reactions to make piperidines and morpholines, the cornerstones of drug discovery.
http://onlinelibrary.wiley.com/doi/10.1002/ejoc.200500389/abstract
Done with surgical precision
I was asked to write a perspective on macrocycles for Chemical Science, which is why I am in the mood for some interesting nuggets this field is loaded with. I hope that the examples I choose will convince the readers that they are in for a big surprise if they think that cyclic peptides are essentially peptides, only cyclic. This is very far away from the truth, but let me use a specific example. What if I gave you a molecule shown below and asked you to invert the stereocenter that is circled from S into R? I cannot see how there would be an intuitively clear answer to this question. However, there is a set of simple conditions that effect this transformation. This chemistry is detailed in a cool paper published in Synlett by Oberhauser and co-workers from Novartis. If you read this work, you will note that there is a clear explanation for why this particular stereocenter is special. By the way, if you feel compelled to share an interesting facet of macrocycle chemistry with me, I will appreciate your insights.
https://www.thieme-connect.com/products/ejournals/abstract/10.1055/s-1999-3103
Strange landscapes
Is there a way to somehow connect the fields of organic chemistry and epigenetics? It seems that these are apples and oranges, but I will try to come up with an angle on this Monday night. Let’s go to the Nobel Prize awarded for Physiology or Medicine in 2012. The recipients, Shinya Yamanaka and John B. Gurdon were rewarded for their contributions to the field of stem cell biology. Here is a “Cliff’s Notes” summary of what Yamanaka did. The ES (embryonic stem) cell corresponds to the top of the “Waddington’s landscape” shown below.
Differentiation into a myriad of cell types found in various tissues is akin to the ball shown above sliding down one of the valleys. The discovery of induced pluripotent stem cells is what Yamanaka’s lab is known for. He and his students found that there is a simple combination of protein factors that can take a differentiated cell (at the bottom of the landscape) and reprogram it back into the pluripotent state on top of the diagram. This was a remarkable discovery no one wanted to believe at the time of its disclosure. It took Jaenisch’s lab to reproduce Yamanaka’s experiments for the research community to buy into induced pluripotency.
A conceptual relationship with organic chemistry would be to consider a bizarre class of reactions with bifurcated transition states. Ken Houk and colleagues published a very interesting review in Angewandte several years ago:
http://onlinelibrary.wiley.com/doi/10.1002/anie.200800918/abstract
Bifurcated reactions are characterized by a divergence of pathways that somehow reminded me of the Waddington’s landscape today. In a particularly thought-provoking paper, Dean Tantillo recently demonstrated that the bifurcation concept is relevant to terpene biosynthesis:
http://www.nature.com/nchem/journal/v1/n5/abs/nchem.287.html
While some might say that this is a rather obscure and esoteric concept, I find this whole bifurcation business to be fascinating. Just think about it: there are reactions lurking out there in which knowledge of the transition state during the rate-determining step is not predictive of the ultimate pathway. So let me ask you: how many times you were “sure” (by doing careful kinetic experiments) about the nature of the slowest step in your reaction and were utterly perplexed when the experimental selectivity was difficult to explain? It does not happen too often, but I think we can all recall some bizarre situations like this. Might we suspect bifurcation in a subset of such occurrences? Let’s face it, more often than not, we have very little clue about the mechanism (recall what George Olah likes to say: “One can never prove the mechanism of a reaction. One can only disprove it”). And now we have bifurcation on top of that? This whole business starts to remind me of Don Rumsfeld’s memorable quote about the “unknown unknowns”:
https://www.youtube.com/watch?v=GiPe1OiKQuk
I am not a fan of this man, but there is something I can take from his statement.
On quantum mechanics
For as long as I can remember, I have always preferred broad concepts over details. My trip to Schrodinger over the past 2 days has been filled with interesting insights, many of which of conceptual nature. I particularly enjoyed some teachable analogies that can serve us well when we explain computational chemistry related material to students. Art Bochevarov of Schrodinger provided a nice way of thinking about the meaning of basis sets and levels of theory in quantum mechanical calculations. I really enjoyed his analogy to cooking. I am sure you are all used to seeing things like “B3LYP/6-31G**” in papers that deal with computation. The B3LYP moniker stands for the level of theory, whereas the 6-31G** portion defines the basis set. The analogy to cooking is in comparing the basis set to ingredients you get at your grocery store (for a given meal, you might want to buy chicken, celery, pepper, salt, coriander). The level of theory is best compared to a cook as this is more or less about which recipe to follow and how to use the ingredients you have in the best possible way. Of course, the dish itself is the final product of a calculation. A lot of care must go into selecting proper ingredients (the basis set). Equally important is the cooking part. If you do things wrong, you might end up with a dish that will have chicken and duck in it, which is not what one wants to see. I think I will always remember this analogy. Among other things in Boston, there were some great insights into how to choose the right basis set for a particular “dish” one wants to cook.
It was interesting to note how many industry folks were at the workshop I attended (everyone but me was from industry). I was trying to see why and apparently there are some compelling reasons to go for high level calculations these days, particularly when it comes to molecular properties such as pKa, logP, and conformational properties. Below is a cool paper that proves the point. This work was mentioned by Art (he also offered some critique of the authors’ basis set/level of theory choices). According to Jaguar, compound 14 has an optimal torsion angle of 0o, which is suboptimal in terms of binding to the pivotal Leu398 of PAK4 kinase (the protein target in this case). With this in mind, a considerably more potent derivative 16 was designed, prepared, and validated. Because of a steric clash, this molecule features the torsion angle of 30o, which enables it to properly interact with Leu398. We’ve got to use more quantum mechanics, ladies and gentlemen.
Student-related matters in Boston
I flew into Boston about an hour ago. My plan is to visit Schrodinger over the next two days (www.schrodinger.com), a software company that delivers innovative solutions in the area of computational chemistry. In our fragment-based work, we rely on their Glide package, which allows us to model some of the scaffolds we pursue against our protein targets in collaboration with the SGC. Having said that, my main interest over the next 2 days is going to be Jaguar, which is a quantum chemistry program I have been interested in for a long time. Pretty much all of my students who use computational tools resort to Gaussian, which is something I myself used back at USC where I was a graduate student between 1992 and 1996. Now that I use Glide for my own docking needs, I consider it more sensible to stick to the same platform, hence my keen interest in Jaguar.
On another note, there are several interrelated themes in what I just wrote. Speaking of fragments, one of my PhD students got a job offer from a very innovative UK company that is active in the area of fragment-based approaches. I will not disclose the names as this is “too hot off the press”, but I did have a shot of brandy with my elated co-worker at 9:30am this morning. Continuing on the theme of my former students, I am off for a dinner at Legal Seafood with Zhi He (currently a PDF at MIT) and Ben Rotstein (currently a PDF at Harvard Medical School). We just got our Chemical Reviews article accepted, which is why we will have some more drinks with Ben as he is the leading author (Serge Zaretsky and Vishal Rai are the co-authors and I tip my hat off to them for a year’s worth of work). I will post a link to this article at some point soon.
To cap off the day filled with interesting student-related matters, just as I was getting off the plane, I got a note from my former student Iain Watson (now at Ontario Institute for Cancer Research) with the following link, which is really perplexing in terms of the story it covers (a possibility that USC will acquire Scripps). But that is not the cool part – what’s really cool is the picture showcasing “the one and only”, my mentor Barry Sharpless, together with Larissa Krasnova, who was my PhD student a while ago (and then did a PDF with Barry):
http://www.utsandiego.com/news/2014/jun/16/USC-Scripps-medicine/
Overall, this has been an eventful day thus far…
Some dazzling aspects of weak interactions
We all know that because of the difference in ground states, diastereoselectivity is inherently more complicated that enantioselectivity. Of course, there are reagent- and catalyst-controlled reactions that override the ground state effects, but they are still largely underdeveloped. In efforts to rationalize and predict reaction outcomes, we are left with various devices that help us build models. When it comes to relative stereochemistry and diastereoselection, organic chemists are the creatures of habit who are trained to recognize conformations and factors that control them. Things such as the Felkin-Ahn model are ingrained in our understanding of reactivity. It makes it ever more amusing when we see exceptions to the rules we are used to, particularly when an example with some ridiculously high level of selectivity comes up. While in France, I heard a great talk by Dr. Kai Rossen from Sanofi-Aventis, detailing the painstaking efforts on behalf of his company to bring artemisinin to market. There are many insightful lessons in the chemistry of this important antimalarial agent. Those of you who know its structure, might think that any attempt to scale up the synthesis of this peroxide is akin to an attempted suicide. Yet, there is a special plant in Italy that handles artemisinin production on a multi-kilogram scale for Sanofi-Aventis. But this will be the subject of a separate post in the future… Today I want to talk about diastereoselectivity. There is one particular step in the synthesis of artemisinin that has some serious “wow” factor attached to it. I refer to the hydrogenation shown below (I drew this based on the data in the paper). This example comes back to my original point about the inherent bias one might expect in a diastereoselective process. It is simply NOT possible to explain the amazing selectivity you see by doodling out some routine rotamers. No way. They are very similar in energy. Instead, the factor that operates in this case appears to be weak non-covalent interactions between the carboxylate group and the olefinic CH bonds of the artemisinate’s cis-decalene system in the transition state. I think I am developing a growing appreciation of weak interactions…
BBC chemistry
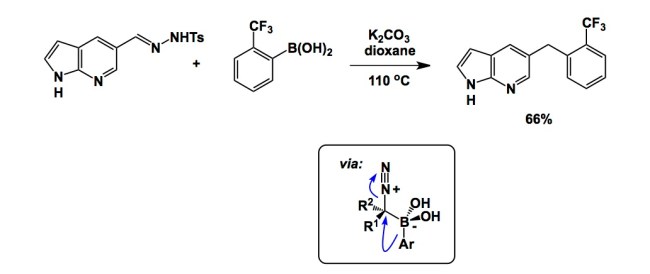
As academic researchers strive to have their methods used in drug discovery, it is important to keep in mind that the reaction scope must include a large proportion of molecules that have a chance to have favourable drug-like properties. This is not always the case, and it is especially true when it comes to carbon-carbon bond forming reactions. Indeed, there are not many methods that accomplish carbon-carbon bond formation without turning synthetic precursors into “grease balls”. And what do you do when you are out of options? You turn to the other extreme and over-emphasize amide couplings, which creates molecules which are not exactly the stallions of drug discovery either.
The paper I intend to discuss is now a couple of years old (see the link below), but there are some practical items of substance in it that deserve attention. As rightly pointed out by Nakagawa and colleagues of Pfizer in their article, published methods tend to focus on excessively lipophilic model substrates, which is not what one wants to see in a molecule that is expected to be cell-permeable and have a chance to go through first-pass metabolism. In their efforts to find a reaction that accepts a broad range of building blocks, tolerates functional groups, and works well in the presence of moisture and air, the authors turned to BBC chemistry (Barluenga Boronic Coupling). This is a fascinating process that involves simple mixing of a boronic acid with a hydrazone. The reaction is reductive in nature and proceeds via an interesting mechanism. I was glad to see this process, originally published in 2009 in Nature Chemistry, in action. Despite the low isolated yields obtained by Nakagawa and colleagues, the simplicity of this protocol is attractive for rapidly assembling relevant molecules.
http://onlinelibrary.wiley.com/doi/10.1002/cmdc.201100339/abstract
Is it weird or what?
At my group meeting earlier today, we were having a problem set related to the FMO theory. This brought to mind a discussion I had with a good friend of mine, Sergey Kozmin, of the University of Chicago (http://kozmin-group.uchicago.edu), who visited our house in Oakville this past weekend. As a matter of fact, this is a surreal coincidence because 24 hours prior to that I saw Vladimir Gevorgyan in Paris, who is also from Chicago (and, like Sergey and myself, hails from the former Soviet Union). In Bill Shatner’s words: “Is it weird, or what?” (https://www.youtube.com/watch?v=MmNgMJWEYJQ).
http://pubs.acs.org/doi/pdf/10.1021/ja00042a049
Continuing along the “weird” angle, I also like when well-known reactions we take for granted behave anomalously. Above is a classic paper by Daniel Singleton that probes the Diels-Alder process with vinyl boranes. Sergey brought this to my attention. What can possibly lurk out there in the old stomping grounds of [4+2] cycloadditions, you might say? Intriguingly, the LUMO coefficients at the vinylic carbon and boron centers are rather close. As a consequence, while the product of the reaction is the expected 6-membered ring, it is the [4+3] transition state that takes hold in this system, defining an interesting secondary orbital interaction. There have been other papers on this subject since Singleton’s report, so I encourage you to look into this literature.
Temporary direction
The fight against entropy is omnipresent in efforts to find new reactions, develop advanced materials and design biologically active molecules. Many of us struggle with this factor and attempt to provide solutions. Take C-H activation as an example. This field has made remarkable strides over the years, but intramolecularity and reliance on the so-called directing groups has been common in finding solutions to some of the longest-standing problems in this field. While attending the FACS meeting in Avignon, I enjoyed listening to the talk by my good friend, Professor Vladimir Gevorgyan of the University of Illinois in Chicago. One of the topics covered in his lecture dealt with silicon tethers that enable the directing element to serve its function and be “erased” towards the end of synthesis using fluoride anion. Alternatively, the C-Si bond could be transformed into something else using cross-coupling or oxidation. The idea of using silicon tether is simple and certainly lifts some of the challenges associated with directing groups that are glued permanently. Below is a link to the paper in Nature Chemistry published by the Gevorgyan lab. There are many other contributions on this topic published by his Chicago team.