Here is a question for tonight: how many cases are out there where a single hydrogen bond dominates the regiochemical outcome of a chemical transformation? Let’s say we are comparing the protio version to its methylated congener, which is arguably the smallest steric perturbation that does not produce a confounding effect. Here is a case from Paul Carlier’s lab published some time ago in Organic Letters. If you look at the two epoxide-containing anilines depicted below, you will note that they differ by a methyl group. Their behavior is strikingly dissimilar, demonstrating how a single hydrogen bond can affect the reactive conformation. The reactions were run neat, by the way. Both outcomes are driven by the logic of trans-diaxial epoxide ring opening, yet the NH-to-O interaction flips the reactive conformation in the first case compared to the second one. If you are looking for a powerful demonstration of the Fürst-Plattner rule, there is probably no better way to show how a relatively weak interaction can control the reaction outcome.
Monthly Archives: February 2017
Detours
I have always been interested in how intermediates with supposedly well-understood behaviour take an occasional detour. At the moment, I am collecting papers of this kind with the goal of writing a review article at some point. There is a lot to talk about here, particularly because the “roads less travelled” are often influenced by subtle structural changes in reaction components. The trouble is that it is not easy to find these cases. Let me illustrate what I mean by using a process you all probably know – the Staudinger reduction of azides. The example below is not new, but is nonetheless instructive because it shows a rare departure from the accepted reaction course. Depending on the group on the phosphorus centre, the reduction of the indole-derived azide proceeds to the well expected iminophosphorane outcome or to the less common triazine heterocycle, whose ring system features three contiguous nitrogen atoms. The mechanism is fascinating, especially if you are programmed to see nitrogen extrusion whenever phosphorus meets azide. I thought this reduction was largely predictable, but I was wrong. Let me know if you are aware of other interesting cases.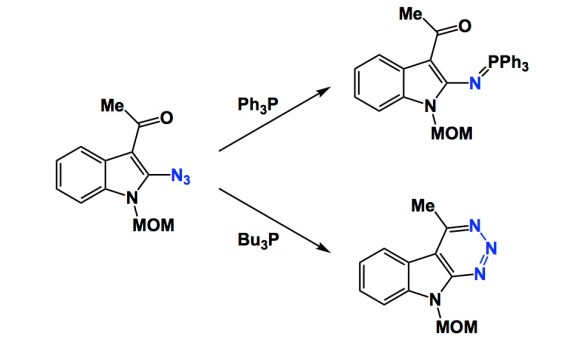
http://www.sciencedirect.com/science/article/pii/S0040402000003227

