I mentioned that my sabbatical is coming to an end in a couple of weeks, which is somewhat sad, given that there are many things I would still like to try. But let’s be honest: things are better left to our graduate students, who will carry the torch forward. As a matter of fact, this was one of my stated goals before I started my protein work at SGC – to try things out, but eventually put it into the right hands. In this regard, it was strategic on my part to stay in Toronto for my sabbatical. I am really happy to see that my student Victoria (shown below) has gained some valuable experience under Elena’s supervision. Victoria now knows the whole process – from cloning to protein production, purification, and crystallization. In addition, Victoria is busy with her synthesis projects. I tip my hat off to her tenacity and ability to pick up new experimental tricks. This is what it’s all about! As for me – it is time to leave protein projects in the hands of experts and not some fluffy amateurs such as myself. I know I will miss my first hand experience, but I will soon need to put back my grant writing/teaching/research advisor hat on.
Monthly Archives: December 2013
Some old insights from crystalline enamines
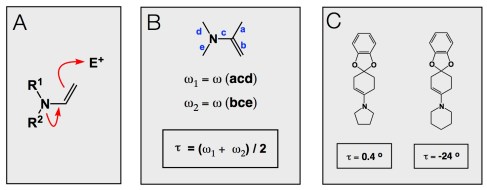
Proline has a number of distinct features. They range from its effect on the formation of turn structures in proteins to proline’s involvement in organocatalytic processes that are rooted in enamine chemistry (inset A). In our lab, we resort to proline when we run peptide macrocyclization: when used as an N-terminal residue in a linear peptide, proline plays an enabling role in the macrocyclization process. We have tried many other secondary amines, yet proline stands out. Interestingly, this observation mirrors organocatalysis in that proline’s core – its five-membered pyrrolidine ring – is a rather special enamine/iminium ion precursor. Notably, piperidine (a six-membered ring) pales in comparison as far as efficiency of iminium ion generation. The other day I was pondering over what makes proline so unique in our reactions and realized that I need to re-read some of the classic papers by Eschenmoser. His excellent Helvetica Chimica Acta work cited below describes X-ray structures of several enamines, including the two shown in inset C. The tau angle is what we want to focus on. Tau is a measure of nitrogen pyramidalization as it describes an average of two dihedrals marked by the small letters in inset B. According to this X-ray study, the pyrrolidine-derived enamine is more or less flat, whereas the piperidine congener is highly pyramidalized. This important crystallographic finding shows that pyrrolidine enamines have a shorter =C–N bond, leading to a higher C-N double bond character compared to their piperidine counterparts. The greater nucleophilicity of pyrrolidine enamines toward C-alkylation becomes clear and also explains why piperidine enamines are more prone to N-alkylation than pyrrolidine enamines.
http://onlinelibrary.wiley.com/doi/10.1002/hlca.19780610839/abstract
Volume: a rarely used parameter
The volume of activation… This is one of those parameters we rarely think about… It represents the difference in the partial molar volume of the transition state compared to that of the reagents. The value of volume of activation provides information about the structural changes that take place in the transition state. Here is a good example: the Baylis-Hillman reaction.
Baylis-Hillman reactions can be really slow (a week to go to completion is not unusual, which is really atrocious). The calculated volume of activation in a prototypical Baylis-Hillman process is approximately -70 cm3/mol. This is a fairly high negative number, indicating a volume reduction when reagents are transformed into the transition state. It can be inferred that an increase in pressure would result in rate acceleration. Indeed, an increase in pressure from 1 bar to 1000 bar provides approximately 15-fold increase in the reaction rate. Please note that no gases are employed here (and pressure still matters)!
One can anticipate that reactions that are accompanied by an increase in the volume of activation should be accelerated when performed in vacuo. Elimination reactions belong to this class. We typically don’t think of vacuum as a means of driving reactions to completion, save for a few rare cases. By the way, volume of activation can be easily measured. Here is a classic paper:
A rare side of boron?
Synthetic chemists are good at creating conditions under which unstable molecules or intermediates can be generated and observed. George Olah’s 1994 Nobel Prize was largely due to his finding that low nucleophilicity media based on antimony chloride provides an environment where carbocations can live long enough to be detected. Under any other conditions, solvents are too nucleophilic to be innocent by-standers.
Now let’s talk about organoboron compounds, which are isoelectronic to carbocations. I just marvel at biological examples that demonstrate how electrophilic functional groups familiar to chemists are prevented from showing their expected properties. Boron in its trivalent form is electrophilic, which is the reason boron inhibitors developed by Anacor work as protease inhibitors. I blogged about these molecules a while back, on August 24. Earlier today I was interested to see a paper describing a recently disclosed Anacor’s structure that defies logic in that it shows a case where boron persists in its trivalent form and is caught in the act of inhibiting a ROCK2 kinase. While the binding mode at the hinge region is common for a kinase inhibitor, I am not aware of a synthetic variant of such hydrogen-bonded pattern involving a boron-containing functionality. As it stands, the fact that boron remains tricoordinate within the kinase active site, is quite unusual.
http://jpet.aspetjournals.org/content/347/3/615.full.pdf+html
Allylic outliers
We all know about allylic strain. Today I want to talk about Inomata’s syn-effect (see the graphic below), which is on display in certain base-promoted isomerization reactions. I have seen Professor Inomata’s thought-provoking work for many years and I think it stands as a very useful example of stereoelectronic balance in acyclic stereocontrol. The peculiar aspect here is that the Z-olefin is favoured. I don’t think cases like this belong to the usual repertoire of topics we discuss while teaching the foundations of acyclic stereocontrol. Indeed, the syn-effect seems to run counter to some of our intuition regarding the build-up of unfavourable steric interactions in transition states. The effect is neat, yet I am kind of happy that there is not too many instances of it out there. I, for one, would not want to mention it when I teach allylic strain. Why confuse all those bright young minds?
In syn-effect, the electronic contribution made when CH sigma orbitals interact with the pi-system, overrides the unfavourable A(1,3) interaction that develops when the R group is forced to be coplanar with the vinyl C-H bond. At least this is what the authors propose, although I have not seen theoretical papers dealing with this phenomenon. It is exceptional cases like this that keep chemistry rolling. There might be other outliers out there and we need to find them.
https://www.jstage.jst.go.jp/article/yukigoseikyokaishi/67/11/67_11_1172/_pdf
Taking risks in chemistry
I just realized that there is something in common between chemical science and economics. Surprised? Hear me out. I will name two important concepts: risk management and exit strategy.
Risk management. When I discuss chemistry ideas with students who are new to research, I often hear questions such as “Do you think this idea will work?”. It takes time to realize that this question has no place in research. This question is irrelevant because nobody has a crystal ball. Very few ideas actually work. The only relevant question is: “Is this idea worth the risk?”. Students who are passionate about science get this really well and are driven to work hard once they realize that the return on their time/effort could be substantial.
Exit strategy. Once we commit to pursuing interesting, yet risky ideas in the lab, we encounter another parallel with economics. I refer to having an “exit strategy”, or abandoning your project because your efforts are best spent elsewhere. In my view, acquiring a good exit strategy is very difficult and I honestly cannot pretend that I have mastered this myself. When do you say that a given research project needs to be stopped? Yet, this knowledge is critical to any research undertaking. I will provide an example that has been articulated by Richard Nisbett, a social psychologist at the University of Michigan (http://www-personal.umich.edu/~nisbett/):
An aging hospital needs to be replaced. Detailed cost estimates suggest that it would be as expensive to remodel this old hospital as to demolish it and build a new one. The proponents of remodelling say that the original hospital was very expensive to build and that it would be wasteful to simply reduce it to rubble. On the other hand, the proponents of building the new hospital say that it would inevitably be more modern. Which path is wiser – remodel the old hospital or build a new one? If you are of the opinion that the old hospital should be remodeled, you have fallen into the “sunk-cost trap” shorthand abstraction (SHA) known to economists. The money spent on the old hospital is irrelevant – it’s sunk – and has no bearing on the present choice. To draw a parallel to research, I submit that this is one of the most difficult lessons to learn when running a lab – how to have an exit strategy and know when to stop pouring resources into a given area. We need to trust our instincts and not fall into the “sunk-cost trap”.
Crush a crystal, hang a drop
I have not written about my sabbatical work for a while. As my sabbatical stint involving protein crystallization approaches the finish line at the end of December, I realize that I have learned a lot of new things. Take, for instance, what we did today with Elena (I blogged about our joint work on several occasions). We recently got some exciting new crystals of a protein I won’t disclose right now (it is just too early in the process). The nature of the protein is not that relevant as I just want to cover an interesting process related to improving crystal quality. We have struggled to crystallize this particular protein for some time. The crystals we have produced prior to today did not diffract well. To improve their quality, we resorted to seeding, which started by us fishing out our imperfect crystals under the microscope, followed by grinding them using a simple device shown on the left side below.
What you see is an Eppendorf vial with a small polystyrene ball in it. When you add to this vial the mother liquor together with your low-quality crystals and vortex them, the ball breaks them up into tiny pieces. Under the microscope, you can actually see them as sharp little fragments suspended in liquid. We made this “crystal slush” last Friday. Today we set up the hanging drops that you see above. Each location is sealed by grease and contains an aqueous buffer (with some additives) at the bottom. Using 0.1microliter volumes you set up hanging drops that have various combinations of the protein of interest and the buffer. Most importantly, to each drop you add 0.3 microliters of the “crystal slush” suspension. That’s it. You do all of this on a thin round piece of glass, invert it, and place it above the buffer solution in each of the 8 locations shown above. Despite gravity, the drops do not fall because of the high surface tension of water… I think we need to take a lot of these protein crystallization tricks into the synthetic organic domain. Personally, I have never tried careful concentration adjustment as a means to drive crystal formation. For Elena, though, there is an enormous difference between 1.25M, 1.3M, and 1.35M protein solutions. Apparently, such spectrum of concentrations can play a huge role in determining the success of crystallization. We’ll see what happens. The main lesson for me is that progress is slow and you just have to be patient. Alas, this is one skill I left behind in my postdoc days… Elena keeps me in check, though!